There is a great deal of hope that Hydrogen is the answer to our dependance on fossil fuels. Unfortunately, this has led to a lot of hype about its use.
Many use cases simply gloss over the very real challenges of this molecule and, judging by many of the concepts, yachting has not been immune to this.
Today, Feadship are building a superyacht with Liquid Hydrogen and Fuel Cells that will allow limited operation without its diesel engines. Whether this remains a one-off example of innovation and ambition, or the future of yachting, is difficult to say given the many challenges that remain.
So, to help get a better understanding of this molecule, I have created the following guide – there is also a video of my recent presentation at the Superyacht Summit Adria at the bottom of the page.
1. Hydrogen

Hydrogen is the first element on the periodic table – the lightest and smallest element known.
It is the most abundant element in the Universe, and on Earth, where it is mainly found in water and hydrocarbons, but extraction requires significant energy.
It’s non-toxic, colourless, odourless and burns with a pale blue flame that is almost invisible in daylight.
Hydrogen is also an indirect greenhouse gas due to reactions with substances such as Ozone and Methane in the upper atmosphere. Studies suggest it has a Global Warming Potential (GWP) over 100 years which is 11x more potent than C02.
At standard temperature and pressure it is 14x lighter than air.
It goes from solid to liquid at -259°C and boils at -253°C – extreme low temperatures.
Hydrogen has excellent gravimetric energy density – the amount of energy by mass. By comparison it contains about 3x more energy than a similar amount of diesel.
So far so good!
Unfortunately, it has poor volumetric energy density – the amount of energy per unit of volume.
Uncontained it requires around 3.5x more space than diesel, but once you factor in the tank, containment room, ventilation, etc., the real figure is over 7x, and compressed is even worse.
The following illustrates how much volume is required for different fuels.
Liquid Hydrogen is the most volumetrically dense form of pure Hydrogen storage, but it would still require a considerable amount of a valuable real estate if you wanted the same range and autonomy of its diesel equivalent.
2. Safety
Hydrogen has been involved in several disasters including the Hindenburg and the Space Shuttle Challenger.
The truth is we know a lot more about the safety of hydrogen these days, though there are still some unknowns, especially with Liquid Hydrogen.
HyResponder a European organisation co-funded by the EU who provide emergency response training to trainers, state:-
Hydrogen is not more (or less) dangerous than any other conventional fuels, but it has a unique set of properties and characteristics.
HyResponder
So what are they?
The first is the flammable range, which is the concentration of Hydrogen in air that will support combustion when ignited by an energy source.
Compared to other fuels Hydrogen has a much larger range of flammability.
It has a very high flame speed 2.65 – 3.25 m/s which is an order of magnitude greater than methane (LNG) or petrol. Hydrogen fires burn quickly but are short lived – the Hindenburg is perhaps a vivid example of this.
Mixtures of between 15 – 59% of Hydrogen in air can be explosive. This is a wider range than other fuels and, under the right conditions, can lead to detonation, where the flame travels with a powerful shock wave at “supersonic speed”.
For comparison, 1 gram of Hydrogen releases 28 x more energy than 1 gram of TNT!
Although it has a high auto ignition temperature of 585°C – the temperature it will ignite without an external source of energy, but requires just 0.3mJ of energy to ignite – static electricity is enough to ignite Hydrogen.
Due to the size of the Hydrogen atom, it can be absorbed into metals. This can cause embrittlement, which can result in material failures. It can also leak through metals and polymers.
Material choice is a key consideration, only those in direct contact, but also those that may be exposed to Hydrogen in the event of a leak.
Hydrogen gas is very buoyant and in the open will quickly diffuse upwards reducing the risk of fire or explosion.
However, compressed Hydrogen will first follow the direction of the high pressure release due to momentum before buoyancy takes over. And, Liquid Hydrogen, due to its extreme low temperature can cause a dense dangerous mixture of Hydrogen at low level.
Liquid Hydrogen expands by 848 to 1, so a small leak can quickly fill a space with a dangerous mix.
Extreme cold temperatures can also distill oxygen from the air. Oxygen itself can be explosive when it comes into contact with organic material such as a bituminous road surface – where you bunker will be important.
In case of a fire, the leak first needs to be isolated before using water as it can excite the flammable mixture.
Key to preventing flammable or explosive mixtures is ensuring the rapid shut down of leaks and effective ventilation.
Hydrogen is unlike any marine fuel in use today, even compared to LNG. There is no doubt safety onboard and in port, both technically and operationally, will be critical.
3. Production Pathways
According to an International Energy Agency (IEA) 2022 report, in 2021 94 Mt was produced mostly by unabated fossil fuels, with less than 1 Mt low-emission Hydrogen.
There are three main production pathways which are denoted by their colour.
- Grey – using fossil fuels, mainly natural gas which emits carbon and methane
- Blue – using carbon capture and storage (CCS)
- Green – using green or low carbon electricity and the electrolysis of water
The molecule is the same whatever the colour, but the emissions are not! Most Hydrogen is used in industrial processes: –
- 40 Mt used in refineries to remove impurities and to upgrade heavy oil fractions into lighter products.
- 34 Mt to produce Ammonia and fertilizer
- 15 Mt to produce Methanol used in chemical industry
- 5 Mt used in the steel industry
Hydrogen is also used in the production of synthetic diesel such as HVO and Power-X fuels such as E-Methanol and E-Ammonia.
There are a significant number of projects in development, especially in countries with abundant green energy e.g. wind, solar and hydro.
Demand is forecast to reach 115 Mt by 2030 and, if all projects in the pipeline were realised by 2030, it would include: –
- 9 – 14Mt of Green Hydrogen
- 7 – 10 of Blue Hydrogen
But, the vast majority would still be Grey
Interestingly, it is expected most Hydrogen will be used in the existing hard to abate sectors, with less than 2 Mt going to go to new uses such as transport.
Water is also key as electrolysis requires around 9 litres of “ultra-pure” H2O for every 1kg of Hydrogen. Water can be sourced from local fresh water supplies, rivers, reservoirs, aquafers, or from desalination of sea water.
The consumption of fresh water, especially in water stressed areas, where many of these new projects are proposed, can impact water for human consumption and agriculture. And, where desalination is used, disposal of the brine into the seawater can have a negative impact on marine life.4. Cost
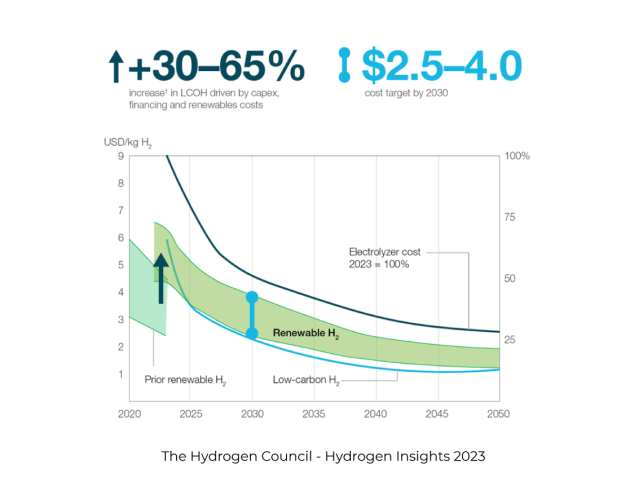
The Hydrogen Council recently released their latest 2030 production cost forecast: –
It is important to note that these are production costs and do not include, compression or liquefaction, storage or transportation.
According to the US Department of Energy 2023 report “Pathways to Commercial Liftoff: Clean Hydrogen” midstream costs i.e. liquefaction or compression, storage, and transport in 2030 could add over $3.00 kg to the cost. And, if a high utilisation (70%) fuel station was included, another $3.60 kg.
Making the price between $5.50 kg truck delivered, and over $10.00 kg from a fuel station – this is still without supply chain margins or taxes.
Today, at the pump in California, you would pay about $36.00 kg for Grey Hydrogen, and in Sweden €36.00 kg for Green Hydrogen.
Once this is adjusted for energy density and efficiency of a marine diesel generator, it’s about 4 – 5x more expensive than diesel.
Blue Hydrogen may offer a cost competitive pathway, however, the WTW emissions would need to be verified and storage guaranteed.
As the Green Hydrogen economy develops, we should see price reductions throughout the eco-system, but it may not be cost competitive in the maritime industry until 2035.
Of, course, the price gap could be closed by subsidies under the US Inflation Reduction Act (IRS), similar EU incentives and/or Carbon Tax, such as EU Emissions Trading System (ETS) for shipping.
5. Storage

There are two main methods of storing pure Hydrogen that is widely used today: –
- As a gas under pressure and,
- as a liquid at low temperature.
It can also be stored in chemicals like Ammonia and Methanol.
There is also ongoing research into storing hydrogen in substances that can absorb and release hydrogen.
These include liquid organic hydrogen carriers (LOHC) and Metal Hydrides. Their main benefit is that they are a safe way of storing Hydrogen at ambient temperature and pressure.
Their downside is the limited amount of Hydrogen stored by weight. And, there is heat produced during hydration, and heat is required to release the hydrogen. This affects overall system efficiencies and cost.
Re-hydration would require discharge of the depleted carrier before bunkering – adding complication, time, and cost to the process.
5.1 Compressed Hydrogen
Hydrogen can be stored in special cylindrical tanks at pressures up to 700 bar.
There are four main types of tanks. Each type uses different materials and construction methods.
- Type 1 – Stainless or aluminium
- Type II – Fibre resin composites with thick metal liners
- Type III – Fibre resin composites with fully metallic wrapped liners
- Type IV – Polymeric liners fully wrapped in fibre resin composites
Due to heating and cooling, a 700 bar tank would need to be over pressurised to 880 bar during filling.
In the EU these tanks need a safety factor of 2.35:1.

You can clearly see from the above a lot of space is lost due to the shape of the tanks – that’s even before the appropriate containment and ventilation is added.
Their use is well established, and generally considered to be a safe way to handle and transport hydrogen.
5.2 Liquid Hydrogen
Liquid hydrogen is carried in insultaed cylindrical or spherical cryogenic tanks at -253 Celsius – a bit like a giant Thermos Flask!
The temperature is maintained by the tank construction, and the initial filing with liquid Hydrogen.

Filling, is a slow process due to temperature differences, and the need to avoid high pressure. Filling too quickly can cause a tank failure, or a gas release to atmosphere.
Like a Thermos, some heat will eventually seep into the tank. This heat energy will be absorbed by the liquid Hydrogen and cause some of it to turn to gas.
This boil-off gas creates pressure which, if not consumed, has to be vented to atmosphere.
For this reason, the maximum capacity is about 85% of the tank volume to allow room for gas expansion. A minimum level is also required to maintain the extreme low temperature.
These two factors limit the amount of usable Hydrogen.
Sloshing, due to a yachts motion in a seaway, can also increase boil-off – even stationary, boil-off can be up to 3% per day, so Liquid Hydrogen is not a great choice for long term storage.
Again, it is widely used, but managing Liquid Hydrogen onboard, and ashore, will be very challenging.
6. Round Trip Efficiency
It’s important to understand that the production, storage and conversion of Hydrogen is energy intensive.
At every stage energy is lost… this can be seen in the diagram below: –
From the initial input 100 units of green energy, you end up with just 26 units of useable energy that is output from a Fuel Cell.
This is why electrification is a better solution for many applications!
7. Energy Conversion
Hydrogen is just a form of energy storage and you still need to convert it into useful energy such as mechanical or electrical.
7.1 Internal Combustion Engines
Apparently it is not difficult to get an internal combustion engine to run on Hydrogen but, getting it to run well, is a different matter!
Engine manufacturers are already familiar with engines running on LNG and are developing Hydrogen engines. Companies include MTU, Caterpillar, MAN and Volvo, though most are being developed for stationary applications like power generation.

Although the combustion of Hydrogen and Oxygen emits only water, air is used, and air is 21% Oxygen and 79% Nitrogen.
At high temperature the Nitrogen reacts to create NOx emissions that are air pollutants and Greenhouse Gases. The amount depends mainly on the temperature of combustion.
There are solutions to reduce NOx emissions, including reducing compression, running a lean mixture, and exhaust gas after-treatment – like an SCR for IMO Tier III.
Hydrogen can also pass into the crankcase where it may ignite. The crankcase needs to be well ventilated and able to relieve pressure.
Due to a “quenching distance” that is less than other fuels, the flame could also get past a partially closed intake valve and cause backfiring.
Then there is efficiency.
For a Hydrogen engine MTU suggests an efficiency of around 42%, which is lower than a fuel cell.
7.2 Fuel Cells
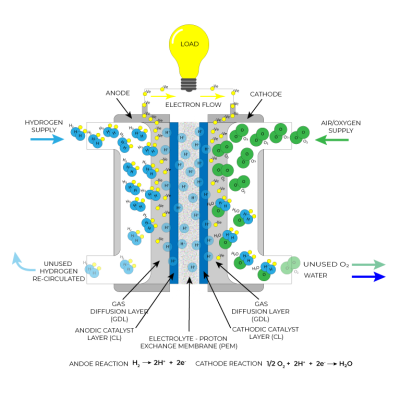
A Fuel Cell is much like a battery where an electrochemical reaction takes place between an Anode and a Cathode submerged in an electrolyte, to produce electricity.
However, unlike a battery that needs to be recharged, a Fuel Cell produces electricity as long as it is supplied with fuel and air.
Although there are different types of Fuel Cell, the most mature and widely used is the Proton Exchange Membrane. These run at low temperatures and have an efficiency of about 55%.
The key parts are the Bi-polar Plate and the Fuel Cell Stack.
- A Cell comprises two Bipolar Plates that sandwich the gas diffusion layer, catalyst and electrolyte.
- Each Cell, when supplied with Hydrogen and air, produces about 0.7 volts.
Multiple Cell’s are joined together in a Stack – much like connecting batteries together in series to increase Voltage.

Apart from electrical energy, the reaction also creates some heat, around 80 Celsius.
The only emissions are unreacted Nitrogen and Oxygen, and water – about 500 litres per hour at 200 kW/h.
As you can see Stack is quite compact, however there are quite a few ancillary parts. These include pumps, control valves, power supply, and ventilation.
I’m standing next to a completed 200 kWh Fuel Cell without its cover plates. And, it’s still pretty compact compared to an equivalent diesel generator.
There are very few moving parts, so maintenance is minimum, meaning less downtime, and lower OPEX.
The critical part is the Stack and is expected to have a life of approx. 30,000 hours at optimal load – at these hours the Fuel Cell efficiency starts to slowly degrade.
Today, a Fuel Cell will cost around 3x more than an equivalent diesel generator.
In the coming years, as use and production scale up, we should see a similar price trend experienced by batteries and solar panels.
8. Conclusions
There is no doubt that Hydrogen has many benefits but, its use in the maritime sector is still in its infancy.
There are still major challenges to overcome, not least the volumetric energy density and how this impacts space.
Although safety is a concern, this will be addressed using a combination of robust risk assessment, design, technology and effective operational procedures, as well as specialist crew training.
Lack of regulations related to the use of Hydrogen as a marine fuel adds complication.
The availability of “local” Green Hydrogen is a limiting factor and is reliant on abundant green or low carbon electricity.
Hard to abate industrial sectors will be first in the queue.
Hydrogen production is capital intensive. Plant utilisation is key to production cost and return on investment (ROI). As is guaranteed off-take.
Today, Hydrogen hardly factors at all in marine fuel forecasts, and that is something to take particular note of as we will be reliant on the scale of commercial shipping for alternative fuel production and infrastructure.
Cost will remain an issue for some years to come, not just the Hydrogen, but also Fuel Cells, related equipment and the complexity of the build.
Liquid Hydrogen is especially challenging to handle onboard and ashore.
Further, yachting is about freedom and spontaneity…it would lose much of its appeal if these were limited by the choice of fuel, never mind the effect on future resale value.
Today, I believe a good case could be made for inland waterways, or ferries. Vessels which have fixed routes, and access to locally produced Green Hydrogen.
But, on balance, and despite the zero carbon promise, I think it’s harder to make a similar case for yachting…perhaps in ten to twenty years?…only time will tell!